From foe to friend: Transforming tumors into personalized vaccines
Radiation has long been a pillar of cancer care, alongside surgery and chemotherapy. But what if it could be used in a new way - to trigger a set of immune responses that transform tumors into killing machines, a type of in situ vaccine?
Silvia Formenti, M.D., has re-visited radiation as a tool that turns tumor from foe to friend. She uses it to help activate the immune system and prime the tumor microenvironment so that the cancer is more responsive to other treatments. In some cases, she has even been able to kill one tumor by irradiating another, inducing what is called the “abscopal effect.”
A native of Milan, Italy, Formenti came to Weill Cornell Medicine and became chair of the newly established Department of Radiation Oncology and Associate Director of Radiation Oncology at the Sandra and Edward Meyer Cancer Center in July 2015. Prior to that, she spent 15 years as the Sandra and Edward H. Meyer Professor and Chair of the Department of Radiation Oncology at New York University Langone Medical Center.
Early in her career, she studied immunology under the tutelage of Malcolm Mitchell, M.D. at the University of Southern California. She learned that an optimal vaccine is: specific to the disease it is designed to treat; sufficiently targeted via neoantigens to prevent toxicity to normal tissues; able to convey robust immune memory; and easy to administer and/or boost.
Radiation can also tick all those boxes.
When Formenti joined NYU, she established a collaboration with a fellow Italian colleague, cancer immunologist Sandra Demaria, M.D. Together they developed the idea that radiation could convert the tumor into a powerful vaccine. But proof would require removal of the strong immunosuppression that characterizes established tumors. The emergence of “immune checkpoint inhibitor” drugs finally made it possible.
An important part of the immune system is its ability to tell between normal cells in the body and those it sees as “foreign.” This lets the immune system attack the foreign cells while leaving the normal cells alone. To do this, it uses “checkpoints” – molecules on certain immune cells that need to be activated (or inactivated) to start an immune response. Cancer cells can escape immune control by using these checkpoints to avoid detection. So immune checkpoint inhibitor drugs were developed to ensure that cancer-killing white blood cells, T cells, can find their targets and mount the body’s natural offensive.
Formenti and Demaria first demonstrated that radiation can help this process along.
“The immune-privileged microenvironment makes it difficult for the immune system to infiltrate. Radiation can break it down and leverage the strength of a patient’s ability to fight their own tumor,” Formenti said.
Ionizing immunotherapy
The ionizing energy released during radiation therapy triggers DNA damage response. It can also alter intercellular signaling, tissue composition and phenotype, and can drive the frequency and diversity of tumors.
“Radiation mimics a viral infection of the neoplastic epithelial cells”, explained Demaria, who has also joined the Radiation Oncology department at Weill Cornell. “It changes the ways cells die, and in doing so, it generates danger signals and enables dendritic (messenger) cells to usher antigens to the surface of tumors, making them visible to T cells. This helps the body overcome at least some of the barriers responsible for T-cell exclusion.”
“Radiation can provide some of the key ingredients to recruit the immune system and convert the irradiated cancer into an effective in situ personalized tumor vaccine,” Formenti said.
Formenti and Demaria have outlined their findings in several academic papers and conference presentation, to great enthusiasm. Their work has opened a new field of research now pursued by many groups nationally and internationally.
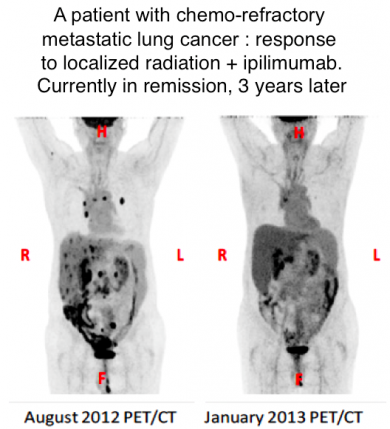
Formenti is currently running four clinical trials that combine immunotherapy and radiation. Some of her results have been astonishing. One lung cancer patient was sent to Formenti for palliative care after failing three rounds of chemotherapy and traditional radiation treatment. His cancer had metastasized to the bones and liver, and his prognosis was poor. Formenti delivered radiation to one of the liver metastases in concert with an immune checkpoint inhibitor that when used alone has little effect: each metastasis went away in a few months and this man is now 48 months disease free with no other treatments.
Crossing hurdles
However, Formenti admits that it would be unfair to highlight just the positives.
“The story is more complicated,” she said.
There are several hurdles to be overcome if radiation is to be applied widely with immunotherapy. The most basic is figuring out the optimal dose and delivery schedule required to cause immunogenic cell death; both seem to be influenced by the microenvironment and the genetic make-up of individual tumors.
Also, immunotherapy has not been effective in all cancers. It relies heavily on the presence of antigens – proteins or other molecules on the surface of cells – that are slightly altered on cancer cells. Some cancer patients don’t have enough antigen-presenting cells to be detected. Or they have other immunosuppressive factors that are difficult to overcome; in some cases, these can actually be exacerbated by radiation.
For example, radiation activates TGFB, a key immunosuppressive cytokine (signaling protein) on many types of cells, notably dendritic cells and T cells. This could actually make the immune system more tolerant of the tumor. To overcome this, Formenti and Demaria worked with Karsten Pilones, M.D., Ph.D., to test combination therapies in which radiation is administered alongside TGFB inhibitor drugs.
Metastatic tumors are especially difficult to treat, and may require special considerations, such as which tumor to irradiate when presented with several sites of metastases. Some organs may be better harnessed; liver lesions, for example, are a very good source of antigens.
“By the time they become metastatic, individual tumors have formed their own unique escape mechanisms,” Formenti said. “If understanding cancer is a challenge, understanding the individualized immune response to cancer is even more of a challenge. Local radiation therapy and multiple immunotherapies may be needed for established tumors.”
Creating a new paradigm
Formenti works with a multidisciplinary team that includes researchers, physicists, technicians and clinicians. Together with other oncologists across disease sites, she provides novel personalized therapies for a variety of cancers at several sites on the Upper East Side and Lower Manhattan, as well as NewYork-Presbyterian/Queens.
They have translated preclinical work into clinical trials in metastatic breast cancer, lung cancer and melanoma, and have also used radiotherapy as an adjuvant to immunotherapy of solid tumors and lymphomas. They are now working to determine best dose combinations, fractionation schedule and patient selection criteria.
“We are changing the radiation treatment paradigm, from ablating the tumor to converting the tumor into an individualized vaccine, in the context of cancer immunotherapy. We are bringing precision medicine to radiation oncology,” Demaria said.
“Our findings have ‘repositioned’ radiation therapy in a novel application, that of an immune modulator. With the advantage of its very focused and localized nature, radiation is ideal for combination with systemic modulations of the immune system” Formenti said. “Immunotherapy is at the forefront of precision oncology, and radiation will play an important role in this process.”
Formenti's immunotherapy clinical trials:
- TGFB blockade and radiotherapy in metastatic breast cancer
- Toll-like receptor (TLR) 7 agonist, cyclophosphamide, and radiotherapy for breast cancer with skin metastases
- Study of REGN2810 (Anti-PD-1) in patients with advanced malignancies
Watch Formenti discussing her work in lung cancer in this OncLive video: